Designing a Digital Experience for Injectable Medications
Snapshot
Client
Fresenius Kabi USA
Company
Multiple, Inc
Focus
Digital Platform Redesign
Role
UX Lead, Designer
Team
Design, engineering
Timeline
6 months to launch
Impact
+114% total sessions
+784% search traffic
+246% referral traffic
+23% average session time
The Problem
Client Description
Fresenius Kabi USA provides medicines, clinical nutrition, and medical technologies for infusion, transfusion, and hospital care. Part of a global network with 85 state-of-the-art manufacturing plants and R&D centers, the company builds on more than 100 years of experience supplying healthcare providers with essential, life-saving therapies.
The Challenge
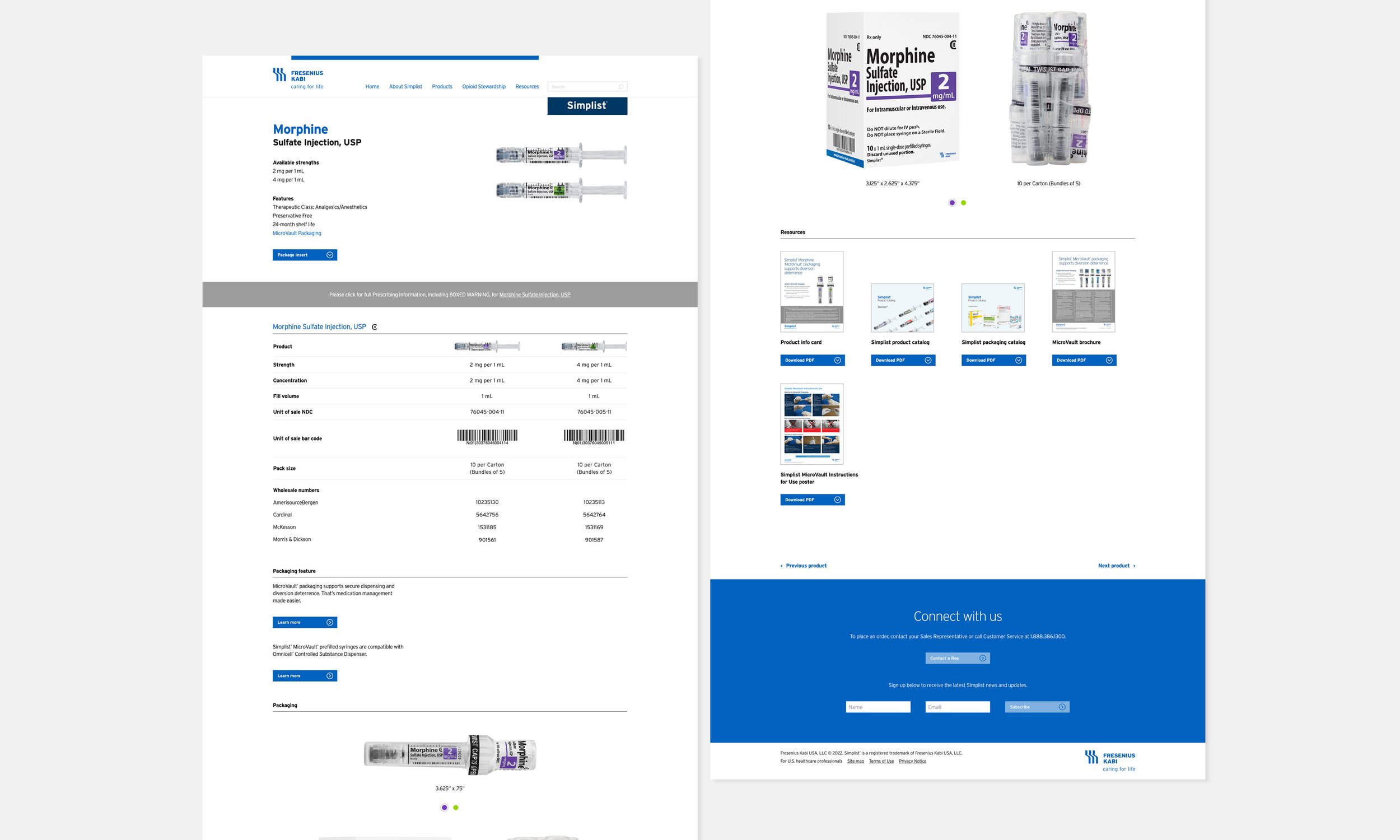
Simplist® is a ready-to-administer prefilled syringe line designed to actively reduce preparation and administration errors. The existing website failed to engage its core clinical audiences due to:
Lack of tailored content for distinct user groups
Unclear messaging around safety and product value
Poor usability and navigation
As a result, critical stakeholders lacked the information needed to make confident, informed decisions at the point of care.
The Reality
Highly regulated healthcare environment
Required rigorous legal and compliance reviews across all content and design decisions.Multiple clinical audiences
Served nurses, pharmacists, and anesthesiologists with distinct needs, workflows, and levels of expertise.Balancing marketing and clinical integrity
Needed to align business goals with clinical accuracy to build trust and credibility.Communicating complex information simply
Translated safety, efficacy, and usage details into clear, digestible content without increasing cognitive load.
Strategy &
Key Decisions
Audience-first content model
Structured the experience around nurses, pharmacists, and anesthesiologists, tailoring messaging and pathways to each group.Clear, safety-driven messaging
Positioned Simplist® as a trusted partner by emphasizing safety, simplicity, and confidence in medication delivery.Scalable, modular framework
Created reusable templates and components to ensure consistency and support future growth.Regulatory-aligned design process
Integrated legal and compliance reviews into the workflow to maintain accuracy without slowing delivery.
My Role
Led end-to-end design, from research through wireframing and final delivery.
Developed detailed wireframes balancing usability with strict regulatory requirements.
Facilitated alignment between design, content, legal, and client stakeholders.
Translated complex medical and product information into clear, actionable digital experiences.
The Impact
+114% total sessions
+784% search traffic
+246% referral traffic
+23% average session time
The redesigned platform significantly improved visibility, engagement, and user understanding, while reinforcing trust with the client and expanding opportunities across additional business units.
Learnings
Align content, design, and compliance early to reduce iteration cycles in regulated environments.
Design for distinct audience needs upfront to avoid retrofitting experiences later.
Discovery & Wireframe Highlights
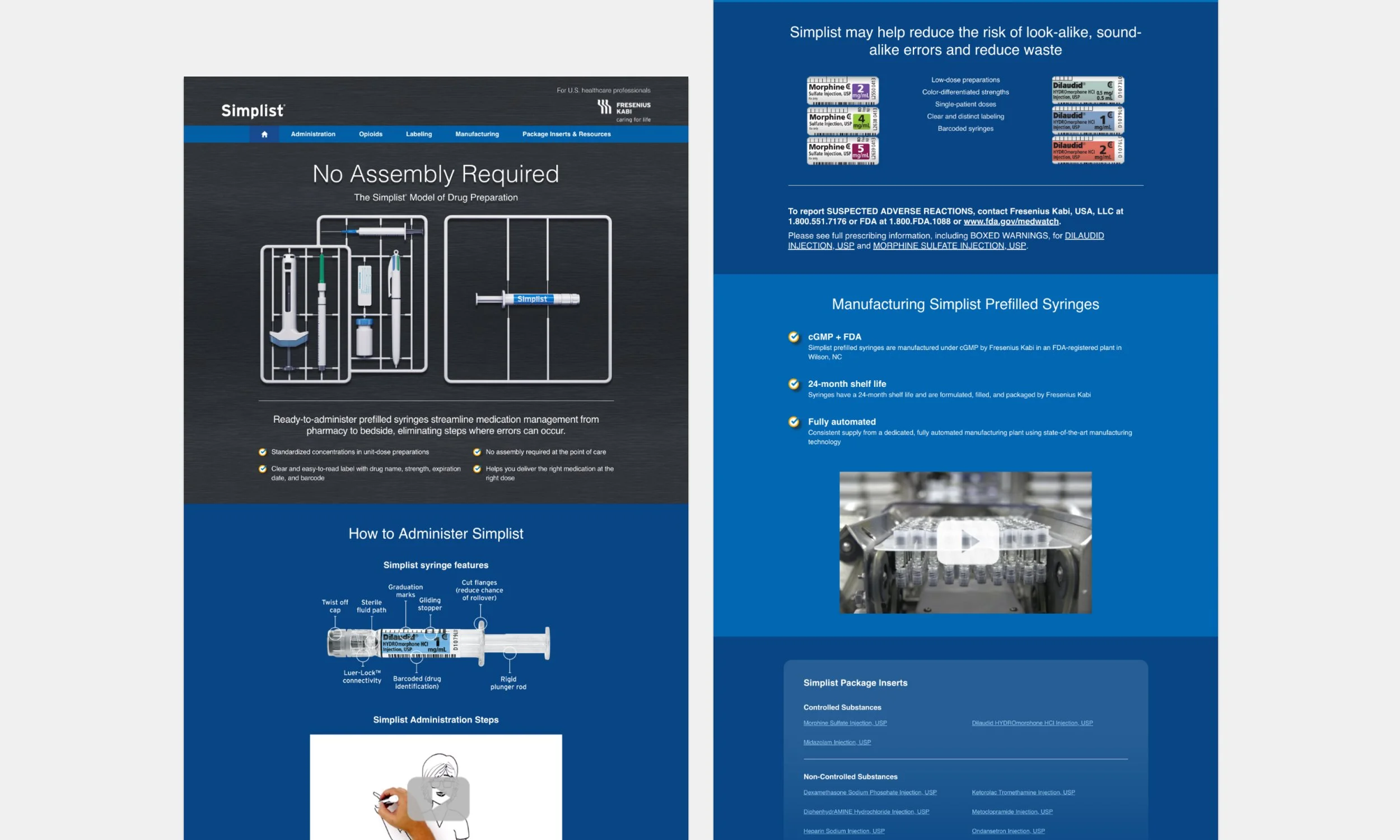
The existing site lacked clear, audience-specific messaging and failed to provide sufficient product, safety, and regulatory information, making it difficult for clinical users to find what they needed. It also underrepresented the brand’s expertise and offered fragmented resources, limiting trust and overall usability.



Key insights from a client working session.


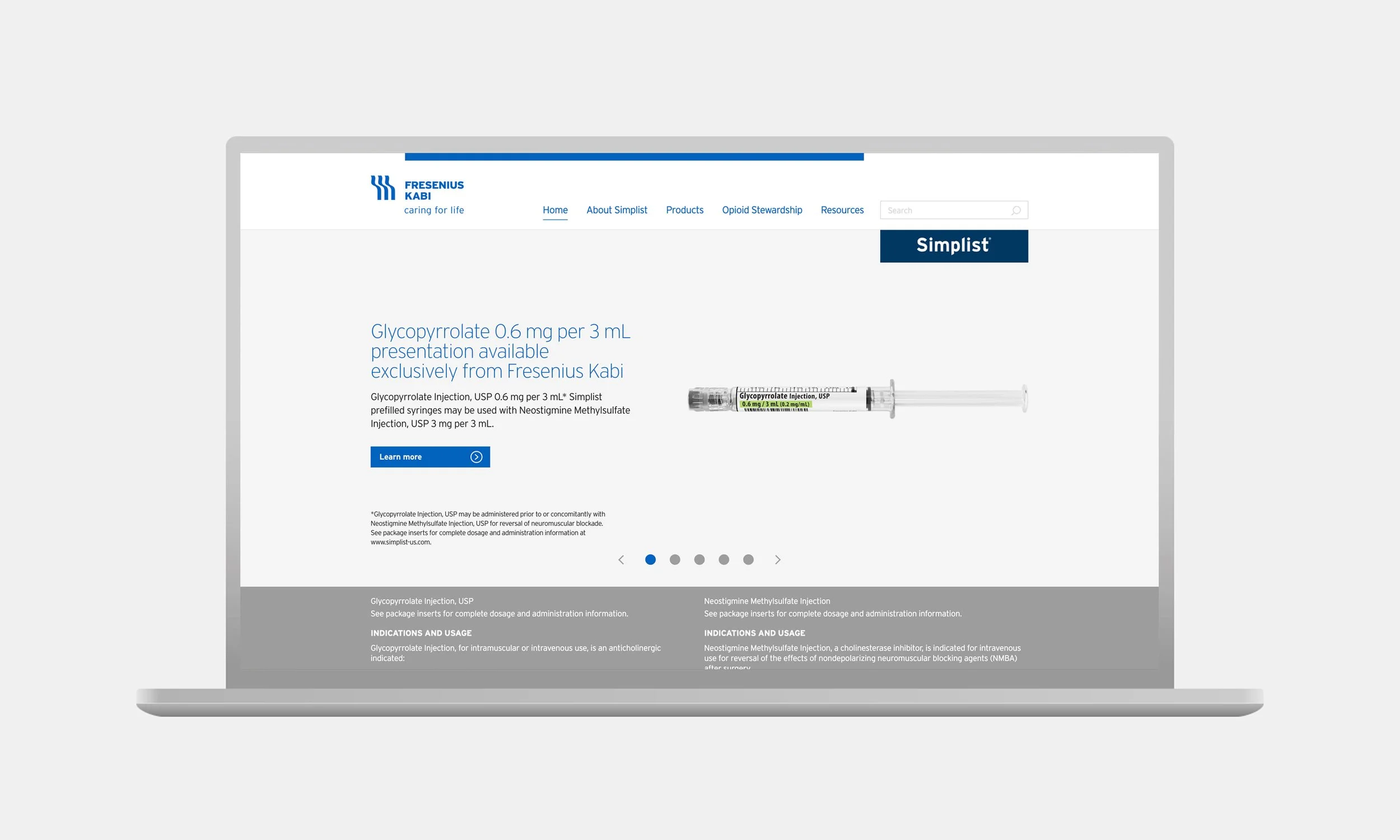
Visual direction
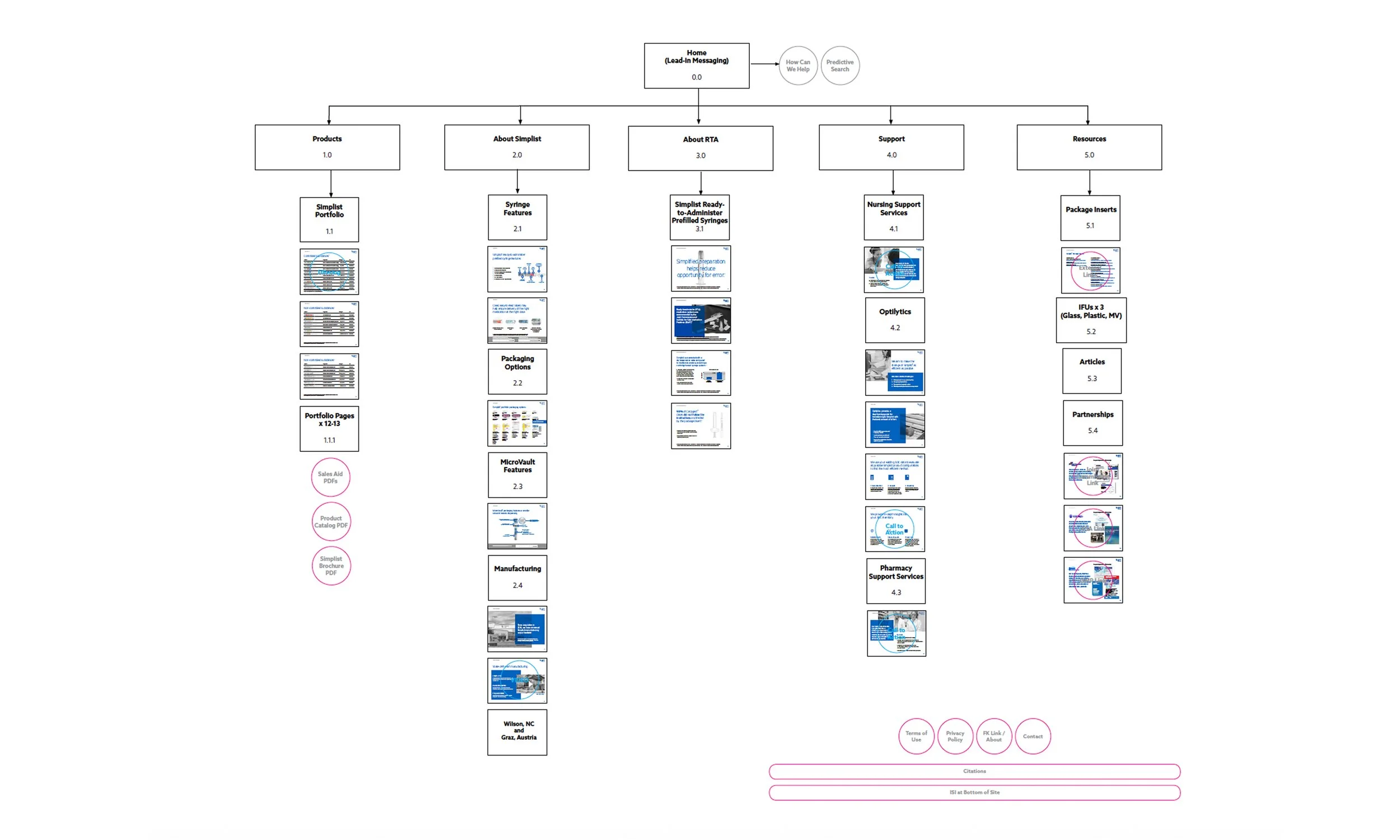
Visual sitemap
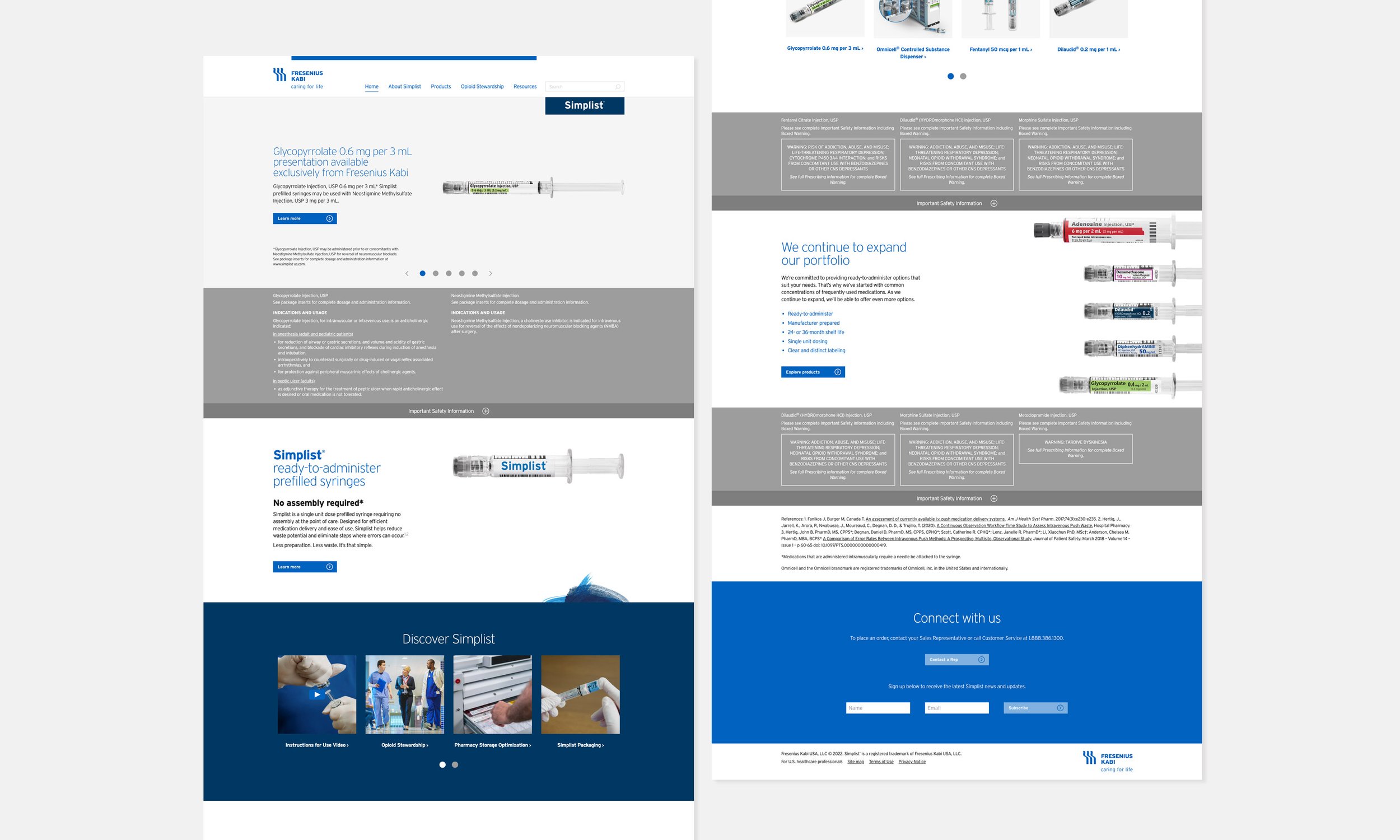
Homepage updated to feature Important Safety Information (ISI) prominently within the product line of sight.
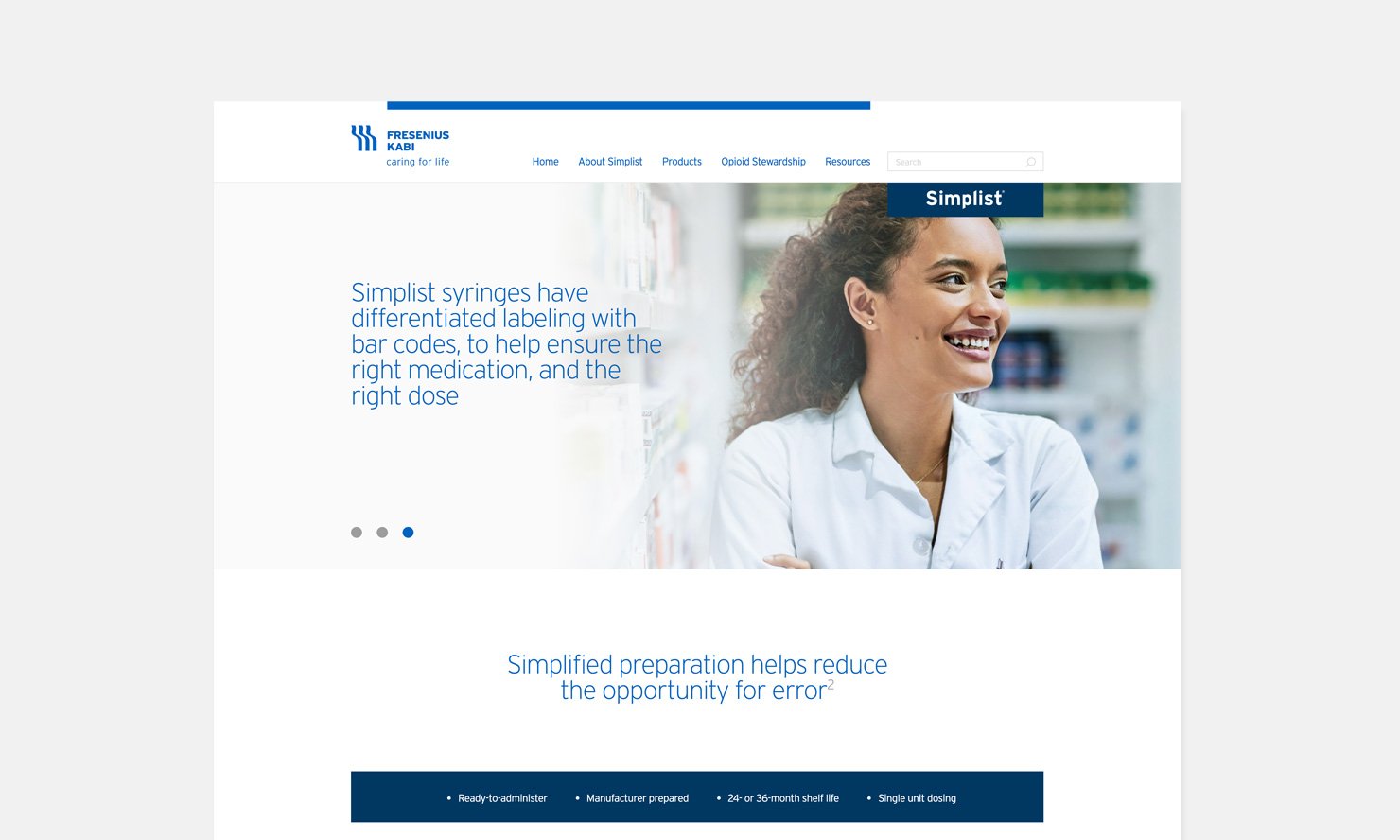
Audience specific content

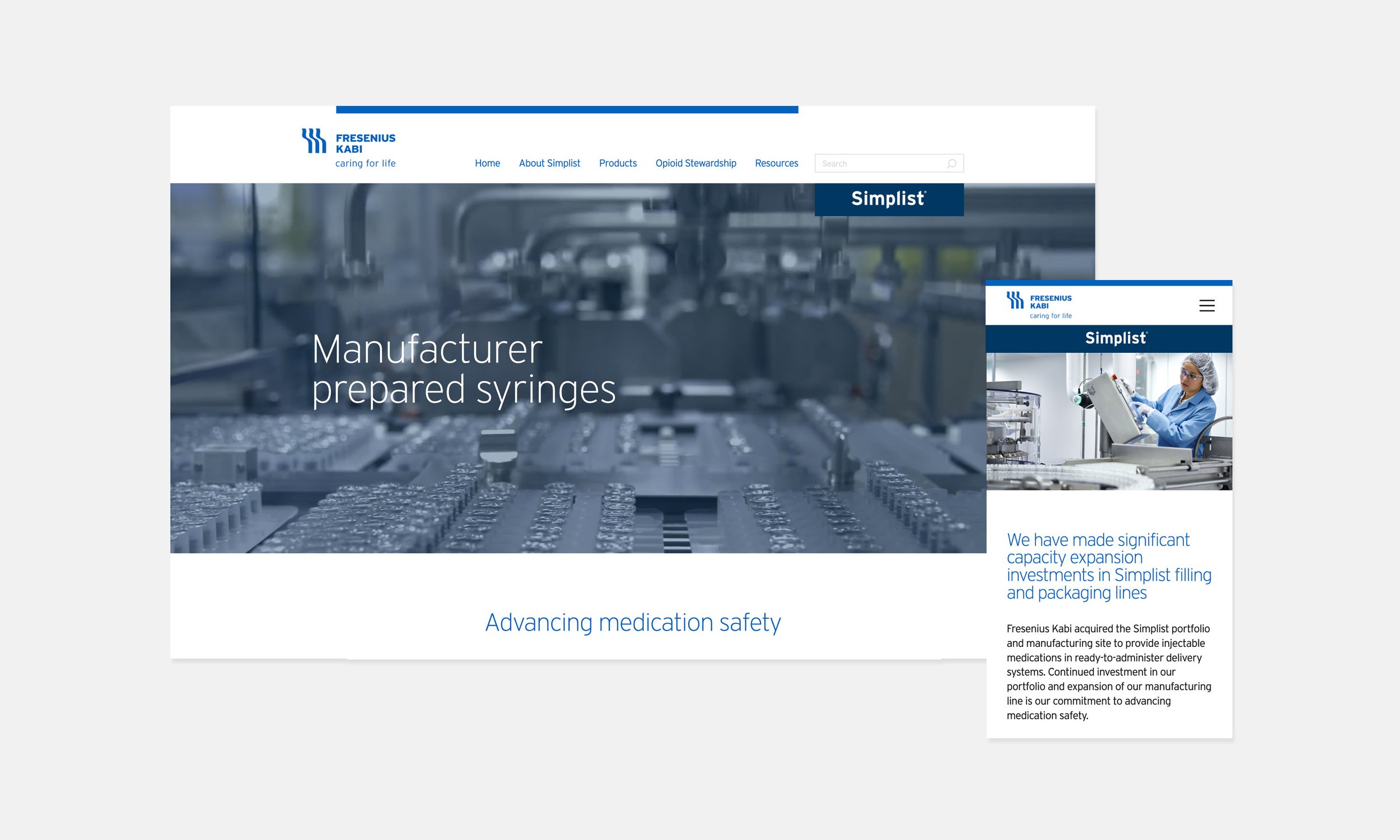
About pages
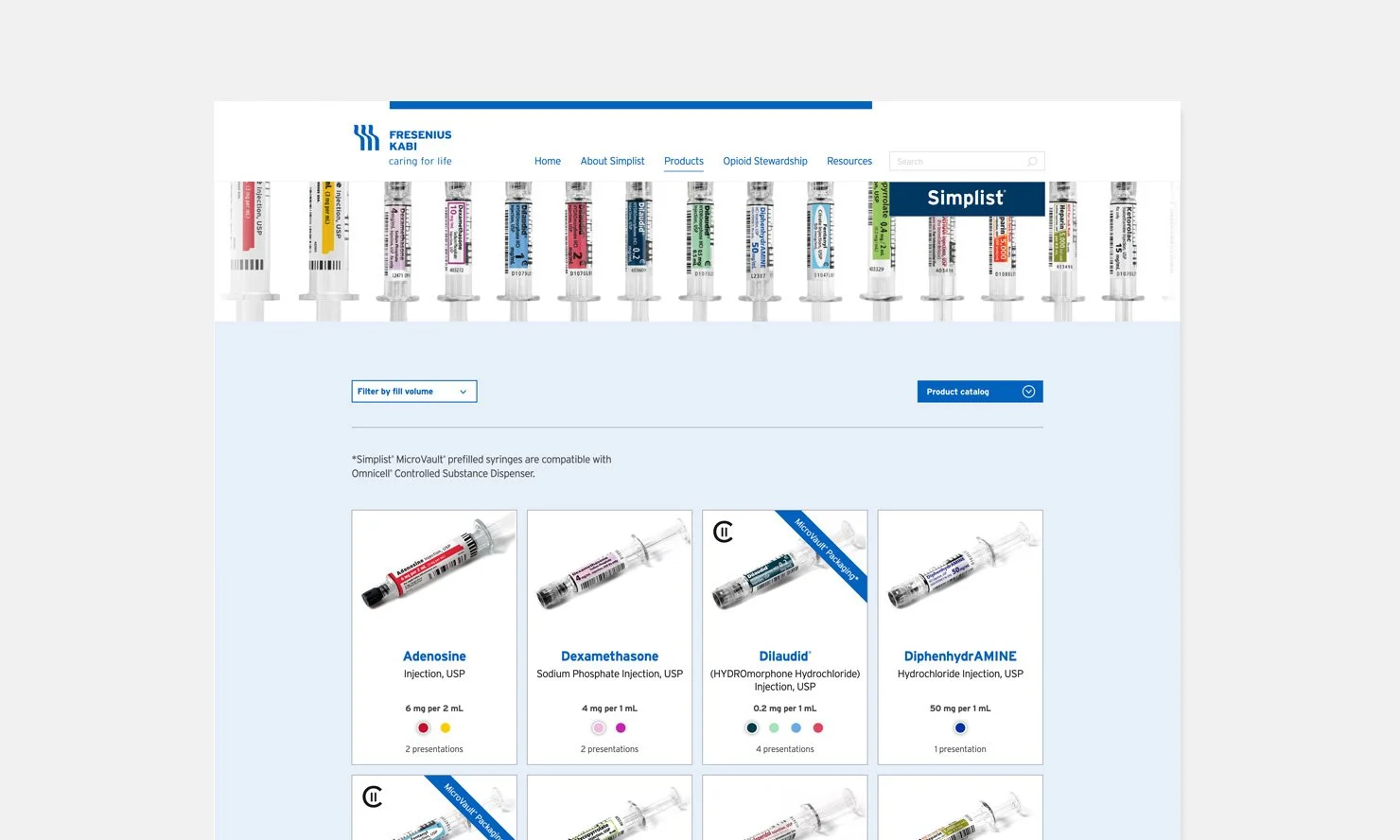
Product portfolio PLP and PDP